査読の際の注意点、チェックポイント: 原著論文
- タイトル
- 著者
- Abstract
- 全体の要約になっているか
- Introduction
- Materials and Methods
- Database and software versions
- Immunohistochemistry
- Real-time PCR
- Regression analysis
- 腸内細菌を扱った論文
- Results: including experimental design
- Discussion
- Reference
- Table
- Figure
広告
タイトル
- タイトルは内容とマッチしているか。
- 個人的には、
タイトルに生物種を含める ことは重要だと考えている。
著者
理想的には、著者の過去の論文を読んだことがある程度には馴染みのある分野の査読を引き受けるようにしたい。
Abstract
全体の要約になっているか
Abstract に論文の内容が過不足なく要約されているかどうか。重要なデータが抜けていたり、全体の conclusion が含まれておらず、データの羅列で終わっているケースもある。Abstract の書き方 も参照のこと。
- General introduction. 1 - 2 sentences.
- Specific introduction, or detailed background. 1 - 2 sentences.
- One sentence clearly stating the general problem being assessed by this paper.
- One sentence summarising the main results of this paper. Started with a phrase like
Here we show... - Result 1, result 2, result 3,... If experiments are organized very well, not many explanatory sentences should be needed.
- One or two sentences to put the results into a general context. Should be closely related to the aforementioned problem.
- Two to three sentences to provide a broader perspective.
Introduction
イントロはレビューではない
Do not describe unnecessary details, which are not related to the contents of the paper, like a review.
Materials and Methods
In this section, authors are requested to provide
Anyway...
現在では、何を論文に記載すべきかをまとめたガイドラインも充実してきている。
対象とする生物種、サンプル数
- 生態学系の論文では、さりげなく複数の種が実験に組み込まれていたりする。Drosophila melanogaster と他の Drosophila など。根拠が明示されていなければ質問する。
- サンプル数は十分か?
- フィールド系の論文の場合、サンプリングの時期は重要。あまりに普遍的な結論を、特定の時期 (夏だけ、など) のサンプルから引き出していないか? 通年サンプリングは基本で、複数年にわたっていればなお良い。
- オスとメス。動物実験をやっていて、性が記載されていなければ、基本的に突っ込んで良い。かなりの項目にバイアスがかかるはずである。
Database and software versions
データベース、ソフトウェアについて書かれている場合、データを得た日付、バージョンなどの情報が記載されているかどうかをチェックする。
Many biological databases, as well as analyzing softwares, are frequently updated recently. Using different version of database and software results in different results. It is necessary to describe the version in M&M section to ensure reproducibility (4).
指摘するときの文例は、例えば
- The authors have used MEGA 7.0 for the phylogenetic analysis. Currently, MEGA 10.0
with advanced functions is available. I would recommend to revise the phylogenetic analysis using the latest version.
Immunohistochemistry
Author guideline package of some journals provide useful information about immunohistochemistry (4).
- Provide the origin (e.g. human, rat, zebrafigh) and sequence of immunogen for antibody.
- If commercial antibodies were used, provide the name of company, immunized species, and monoclonal/polyclonal.
- Characterization information such as western blot, radioimmunoassay and ELISA should be included. This is because it is difficult to find non-specific bindings in immunohistochemistry.
- Negative controls: pre-adsorption with the original antigen, and/or incubation with secondary antibody only.
- If transfected cells are used, two negative controls are required. Non-transfected cells stained with primary and secondary antibodies, and transfected cells stained with secondary antibody only.
Real-time PCR
To be updated. See minimum information for the publication of real-time quantitative PCR experiments guidelines for now.
1.2 倍とかのわずかな差で、とくに異なる遺伝子の mRNA 量を比較している場合には注意が必要。プライマーの増幅効率の差を考えると、そこまでの詳細な比較は原理上できないと思われる。
Regression analysis
If the paper performs regression analyses, check the following points.
- Is the selection of function (linear, exponential, logarithmic etc) appropriate? Computers can calculate how their data fit to the specific function, but authors determine functions to calculate.
- Are the coefficient of determination and statistic results shown?
腸内細菌を扱った論文
サンプルが病気をしていないか、抗生物質の投与歴がないか。
Results: including experimental design
Time- and concentration-dependency?
- Dose と time を振る。
- タイムコースの場合、control は Time 0 だけではなく、全ての time points についてとるべき。
- Solvent を使っている場合、control は未処理のものと solvent control の両方をとるべき。
If the paper examines the effect of some reagents on animals or cells, the experiment should test dose- and time-dependency in general.
A good example is the this Figure (1). If you have only one timepoint, for example 1 h, you would conclude that "Bcl-2 protein expression was significantly increased."
However, if you have only the 12 h result, you should conclude in the other way; "Bcl-2 was decreased". Analyzing limited timepoints generally have a risk to miss important results.
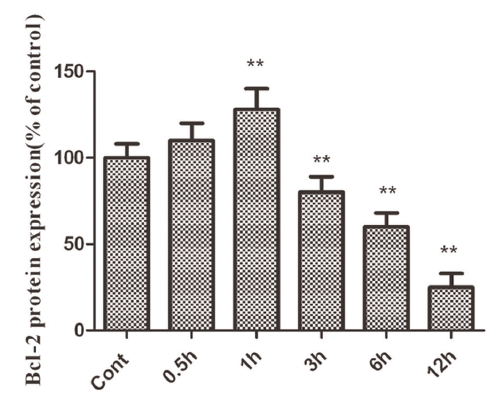
Bar graph
- Scale, X and Y labels are OK?
- Error bar, SD or SE? N is mentioned in legend?
If the value is normalized, the data should be carefully checked.
Statistics
Pointing out statistical mistakes is often difficult because reviewers should know well about statistics. I hope that this page helps Japanese people to learn statistics.
- Duncan's multiple-range test is now widely criticized because of its poor control over type I error. The *** test is a better option.
統計表現の書き方 のページでもまとめたが、
Discussion
モデル図を最後の図として出しているが、論文の結果が少ししかその図に含まれておらず、壮大な絵を書いているというのはありがちなミス。
Figure
Figure legend
Figure legend is getting longer and longer nowadays. Many papers mention results in figure legends. Alothough there are many styles, in my opinion, the optimum length of legend is
You might want to point out this if the paper does not include the following information:
- Number of samples (N)
- Meaning of error bars. Standard error or standard deviation.
- Meaning of asterisks. Typically P < 0.05.
- Abbreviations used in Figures
広告
References
Cui et al. 2012a. Propofol prevents autophagic cell death following oxygen and glucose deprivation in PC12 cells and cerebral ischemia-reperfusion injury in rats. PLoS ONE 7, e35324.- How to construct a Nature summary paragraph. Link.
- Obokata et al. 2014. Essential technical tips for STAP cell conversion culture from somatic cells. Protocol Exchange. doi:10.1038/protex.2014.008
- Author information pack. General and Comparative Endocrinology.
コメント欄
サーバー移転のため、コメント欄は一時閉鎖中です。サイドバーから「管理人への質問」へどうぞ。
